Autoimmune hemolytic anemia (AIHA) is a rare blood disorder where the immune system mistakenly attacks and destroys red blood cells, leading to anemia1 . This condition affects approximately 1 to 3 people per 100,000 each year and can cause symptoms ranging from mild fatigue to severe organ complications1 2. AIHA is classified by the type of antibodies involved and whether it occurs independently or with other diseases, which influences treatment and prognosis3 4.
Types of Autoimmune Hemolytic Anemia
AIHA is mainly classified by the temperature at which the autoantibodies react and by whether it is primary or secondary to other conditions3 .
Warm Autoimmune Hemolytic Anemia
Warm autoimmune hemolytic anemia (wAIHA) is the most common form, accounting for about 70% to 80% of adult cases and roughly half of pediatric cases5 6. It involves IgG autoantibodies that bind to red blood cells at normal body temperature (around 37°C), leading to their destruction primarily in the spleen7 8. This extravascular hemolysis causes premature red blood cell (RBC) removal, resulting in anemia7 .
wAIHA can be idiopathic (primary) or secondary to autoimmune diseases, infections, cancers, or drugs3 7. The disease progression is often gradual but can sometimes be rapid and severe9 .
Cold Autoimmune Hemolytic Anemia
Cold autoimmune hemolytic anemia, also known as cold agglutinin disease (CAD), involves IgM autoantibodies that bind to RBCs at cooler temperatures, typically below 30°C (86°F) 1011. These antibodies activate the complement system, leading to red blood cell agglutination and destruction, often triggered by cold exposure10 8.
CAD mainly affects adults aged 40 to 80 years and accounts for about 10% to 20% of AIHA cases11 2. A rare subtype, paroxysmal cold hemoglobinuria (PCH), is characterized by a cold-reactive IgG antibody causing intravascular hemolysis after cold exposure, mostly in children12 .
Primary and Secondary Autoimmune Hemolytic Anemia
AIHA is further classified as primary (idiopathic) when no underlying cause is identified, accounting for approximately half of cases3 7. Secondary AIHA occurs in association with other conditions such as autoimmune diseases (e.g., lupus, rheumatoid arthritis), infections (e.g., Epstein-Barr virus), lymphoproliferative disorders (e.g., lymphoma, chronic lymphocytic leukemia), or drug exposure3 7131415.
| AIHA Type | Characteristics | Common Associations |
|---|---|---|
| Primary AIHA | No identifiable cause; idiopathic | - |
| Secondary AIHA | Linked to other diseases or drugs | Autoimmune diseases, infections, cancers, medications3 71314 |
AIHA Symptoms and Signs
Symptoms of AIHA vary widely depending on the severity and type of hemolysis16 2. Many patients experience gradual onset of anemia symptoms, while others may have acute presentations.
Common symptoms include:
- Fatigue and weakness due to reduced oxygen delivery16 2
- Pale skin and mucous membranes2
- Rapid heartbeat (tachycardia) as the body compensates for anemia2
- Shortness of breath and chest pain in severe cases2
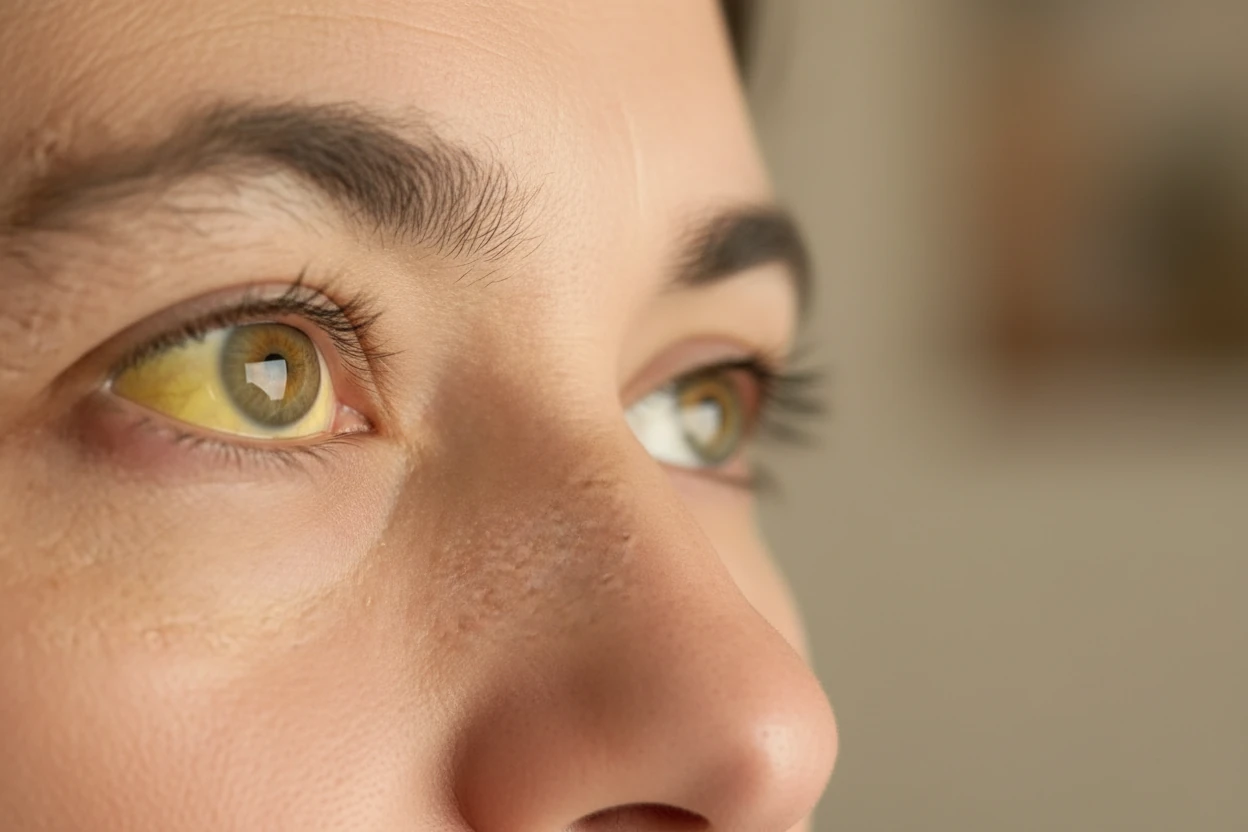
- Jaundice (yellowing of skin and eyes) caused by increased bilirubin from RBC breakdown16 2
- Dark-colored urine from hemoglobinuria2
- Enlarged spleen (splenomegaly) causing abdominal fullness or discomfort17 16
Cold AIHA may present additional symptoms related to cold exposure:
- Cold hands and feet with bluish or reddish discoloration2 10
- Pain in extremities and possible skin ulcers or necrosis in severe cases10
Cognitive difficulties such as brain fog can also occur due to anemia-related reduced oxygen delivery to the brain18 .
Causes and Risk Factors
AIHA results from the immune system producing autoantibodies that mistakenly target red blood cells, leading to their premature destruction7 1. The exact cause of primary AIHA remains unknown, but secondary AIHA is triggered by various conditions.
Risk Factors
- Autoimmune diseases such as systemic lupus erythematosus, Sjögren's syndrome, systemic sclerosis, and rheumatoid arthritis13 3
- Infections including influenza, pneumonia, tuberculosis, Epstein-Barr virus, cytomegalovirus, and varicella19 32
- Hematologic malignancies like lymphoma and chronic lymphocytic leukemia, as well as some solid tumors14 203
- Certain medications, including NSAIDs, chemotherapy agents, antivirals, and antibiotics15 3
- Immunodeficiency states, including post-transplantation immunosuppression21 3
- Cold exposure as a trigger in cold AIHA10
The interplay of genetic predisposition, immune dysregulation, infections, and environmental factors contributes to AIHA development22 .
Diagnosis and Testing
Diagnosing AIHA involves clinical evaluation and laboratory testing to confirm hemolysis and detect autoantibodies16 4.
Key diagnostic tests include:
- Complete Blood Count (CBC): Measures red blood cell count, hemoglobin, and other blood components; typically shows normocytic anemia23 18
- Reticulocyte Count: Indicates bone marrow response; elevated in hemolysis18
- Peripheral Blood Smear: Reveals morphological changes in RBCs supporting hemolysis18
- Direct Antiglobulin Test (Coombs Test): Confirms immune-mediated RBC destruction by detecting antibodies or complement on RBC surfaces16 7
- Lactate Dehydrogenase (LDH): Elevated due to RBC destruction18
- Haptoglobin: Decreased as it binds free hemoglobin released from destroyed RBCs18
- Indirect Bilirubin: Increased from hemoglobin breakdown18
- Cold Agglutinin Titer: Helps diagnose cold AIHA by measuring cold-reactive antibodies10
Additional tests may be performed to identify underlying causes, such as autoimmune panels, infection screening, and cancer workup7 3.
Treatment Options
Treatment of AIHA aims to reduce immune-mediated red blood cell destruction, alleviate anemia, and manage underlying conditions8 16.
Medications
- Corticosteroids: Prednisone is the standard first-line therapy for warm AIHA, reducing autoantibody production and hemolysis; about 70-80% of patients respond within weeks16 824
- Rituximab: A monoclonal antibody targeting B cells, used as second-line therapy for steroid-refractory warm AIHA and first-line for cold AIHA, often combined with other immunosuppressants25 2311
- Immunosuppressants: Drugs like azathioprine and cyclophosphamide may be used in refractory cases to suppress the immune response26 8
- Complement Inhibitors: Newer agents such as sutimlimab target complement activation in cold AIHA, reducing hemolysis26 11
- Intravenous Immunoglobulin (IVIG): Occasionally used in severe cases to modulate immune activity26
Splenectomy
Surgical removal of the spleen is considered for refractory warm AIHA cases16 . The spleen is a major site of antibody-coated RBC destruction and antibody production8 . Splenectomy can induce remission in many patients but carries risks such as infection and surgical complications16 27.
Blood Transfusions
Transfusions are used in severe anemia to restore oxygen-carrying capacity temporarily18 16. They do not treat the underlying cause but provide critical support during acute hemolysis or treatment delays9 .
Lifestyle Adjustments
- Avoid cold exposure in cold AIHA to prevent antibody activation and hemolysis10 3
- Manage underlying conditions such as autoimmune diseases or infections to reduce AIHA flares13 16
- Monitor and maintain vaccinations to reduce infection risk, especially after splenectomy or immunosuppression16 2
AIHA Prevention Strategies
Preventing AIHA is challenging due to its multifactorial causes8 . However, managing triggers and associated conditions can reduce the risk of disease flares:
- Control autoimmune or neoplastic diseases linked to secondary AIHA13
- Prevent infections through vaccination and hygiene measures16
- Avoid cold exposure in cold agglutinin disease10
Early diagnosis and treatment improve outcomes and may normalize life expectancy in many patients28 .
Potential Complications
AIHA complications arise from anemia itself and from treatments used to control the disease16 .
Common complications include:
- Splenomegaly: Enlarged spleen can cause pain and risk of rupture17
- Infections: Immunosuppression and splenectomy increase susceptibility to bacterial infections16
- Thrombosis: Hemolysis is a risk factor for venous and arterial blood clots, including pulmonary embolism and myocardial infarction29
- Ischemic Skin Ulcers: Particularly in cold AIHA, due to impaired blood flow in extremities10
- Renal Impairment: Hemoglobinuria and iron deposition from hemolysis may damage kidneys16
- Organ Failure: Severe anemia can precipitate liver or cardiac failure if untreated17 16
Summary and Key Points
“Autoimmune hemolytic anemia is a group of disorders characterized by a malfunction of the immune system, resulting in the production of autoantibodies that attack red blood cells as if they were substances foreign to the body.”
— Gloria F. Gerber, MD, Johns Hopkins School of Medicine, Division of Hematology9
Autoimmune hemolytic anemia is a rare disorder where the immune system attacks red blood cells, causing anemia and related symptoms1 . It is classified mainly into warm and cold types based on antibody characteristics and temperature sensitivity3 . Diagnosis relies on blood tests confirming hemolysis and immune involvement16 . Treatment includes corticosteroids, immunosuppressants, rituximab, splenectomy, and supportive care such as transfusions and lifestyle changes16 25. Complications can be serious but are manageable with prompt and appropriate care16 29.
Key Takeaways:
- AIHA involves immune-mediated destruction of red blood cells, leading to anemia and systemic symptoms1 7.
- Warm AIHA is the most common type, caused by IgG antibodies active at body temperature5 .
- Cold AIHA involves IgM antibodies activated by cold exposure, causing complement-mediated hemolysis10 .
- Diagnosis requires a combination of clinical evaluation and laboratory tests, including the Coombs test16 .
- Treatment is tailored to AIHA type and severity, with corticosteroids as first-line for warm AIHA and rituximab for cold AIHA16 25.
- Prevention focuses on managing underlying diseases, avoiding cold exposure, and infection control8 10.
- Complications include splenomegaly, infections, thrombosis, and organ damage, emphasizing the need for careful monitoring16 29.
“AIHA is fundamentally orchestrated by a complex interplay between innate and adaptive immune components, including autoreactive B and T lymphocytes, macrophages, and the reticuloendothelial system. Central to disease pathogenesis are two interrelated mechanisms: clonal B-cell expansion with autoantibody production and complement activation.”
— Alessandro Costa, Department of Medical Sciences and Public Health, University of Cagliari, Hematology Unit, Businco Hospital, ARNAS Brotzu, Cagliari, Italy22
Frequently Asked Questions
What causes autoimmune hemolytic anemia?
AIHA is caused by the immune system producing autoantibodies that attack red blood cells. It can be primary (idiopathic) or secondary to autoimmune diseases, infections, cancers, or medications3 7.
How is AIHA diagnosed?
Diagnosis involves blood tests such as complete blood count, reticulocyte count, peripheral smear, and the direct antiglobulin (Coombs) test to detect immune-mediated hemolysis16 18.
Can AIHA be cured?
There is no complete cure for AIHA, but it can be effectively managed with medications, surgery, and supportive care. Early treatment improves prognosis and quality of life2 16.
What are the main treatment options for AIHA?
Treatment includes corticosteroids, rituximab, immunosuppressants, splenectomy, and blood transfusions depending on the AIHA type and severity16 25.
Can AIHA be prevented?
Prevention is difficult, but managing underlying conditions, avoiding cold exposure in cold AIHA, and preventing infections can reduce the risk of flares8 10.